Molecular Tools to Address Alzheimer's Disease
Project Team
Team profile by members of the Gene Therapy for Alzheimer's Disease project team
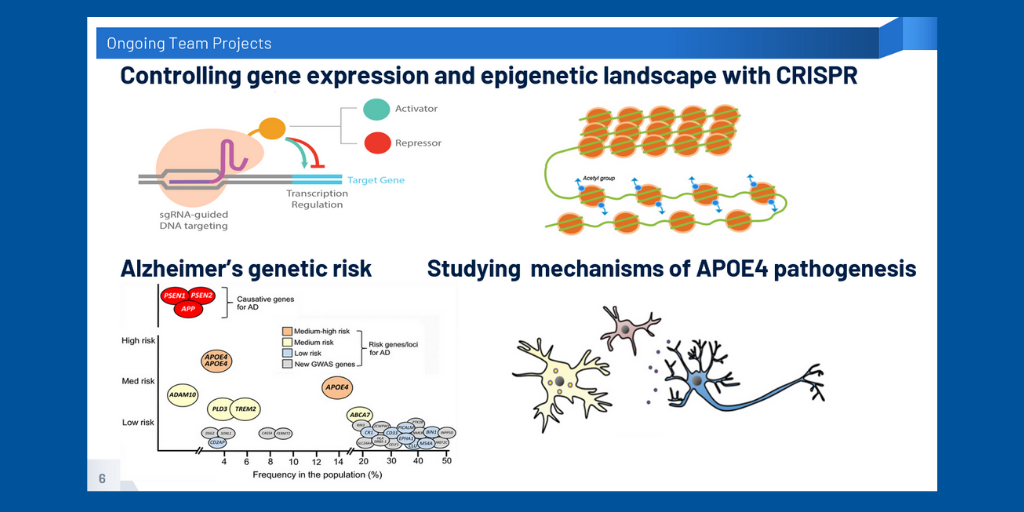
Our team consists of three distinct yet intertwined interdisciplinary groups. In the Chiba-Falek Lab (Duke School of Medicine, Neurology Department, Translational Brain Sciences Division) we’ve been investigating the role of apolipoprotein E (APOE) and its different isoforms in late onset Alzheimer’s disease (LOAD) pathology. Assessing neurons, astrocytes, and microglial-like cells, our human-induced pluripotent stem cell (hiPSC) derived model has been able to substantiate phenotypic differences between APOE ε2, ε3, and ε4 alleles, suggesting a role for APOE in the maintenance of the nuclear envelope integrity that may lead to neuronal loss and changes in cell homeostasis. We have also created polygenic risk scores that can be used to predict the likelihood of developing LOAD and the likelihood of depression among LOAD patients. In our efforts to characterize the heterogeneity of AD, we’ve explored the need to refocus on targets like APOE and shift the paradigm of LOAD drug development toward precision medicine - in a way that considers LOAD patients’ genotypes and phenotypes.
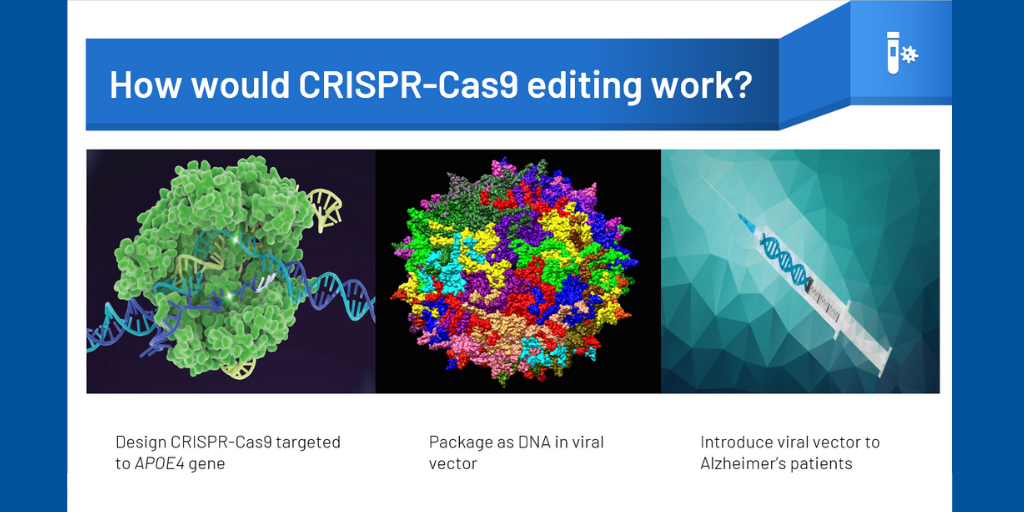
In the Kantor Lab (Duke School of Medicine, Neurobiology Department), we’ve focused on designing the molecular tools necessary to silence gene expression of APOE4, using CRISPR-Cas9 technology and lentiviral vectors. After results from previous experiments using a series of vectors containing small guide RNAs (guiding our vector to the APOE promoter) and DNMT3A as an effector (used to control levels of DNA expression by adding chemical tags) suggested that we were able to achieve successful downregulation of APOE4, our focus this semester was on ways to further refine the specificity of our design. In addition to generating new cell lines of various genotypes to provide a model system for future experiments, so far we’ve explored ways to use inducible systems for more precise temporal and spatial control and how to best optimize size efficiency of AAV viral vector delivery in order to create more viable and effective forms of gene therapy.
In an effort to explore future challenges associated with finding a disease modifying treatment, or DMT, for LOAD - which seeks to stop or slow the progression of a disease by making alterations to the pathological steps leading to its onset - we’ve also been working in collaboration with Dr. Angrist (Initiative for Science and Society) to trace the development of earlier gene therapies. This research emphasizes the importance of both reflecting on the past and preemptively reimagining healthcare delivery, in order to address issues such as pricing, cost, treatment, and patient stratification, rather than waiting until a successful DMT becomes a reality. Most recently, we have considered multiple sclerosis as a crystal ball of what Alzheimer’s patients may encounter once a DMT enters the market.
As a group, we faced challenges in compiling the foundational knowledge necessary to grasp not only a thorough comprehension of Alzheimer’s disease itself, but specifics of the more specialized areas of research each team member contributed to the project as well, given our diverse academic backgrounds. Along with limitations to in-person lab experiences due to COVID-19, we were also challenged to think outside the box, given that very little pre-existing literature exists regarding the development of DMTs for AD, requiring us to piece together elements of existing models with our own novel therapies in order to imagine such a concept.
Group successes included our ability to integrate the three individual portions of our project towards our common goal of ultimately combating the ever-growing prevalence of AD through successful implementation of a DMT. Frequent group presentations and joint lab meetings encouraged open communication and collaboration, enabling us to better understand each other's unique areas of focus, and increased our appreciation of the integral relationship between science and policy - knowing that one cannot exist in a vacuum without the other.
Gene Therapy for Alzheimer's Disease and Ethical Aspects of Genome Editing
Poster by Angela Wei, Anna Yang, Ishika Gupta, Mohanapriya Cumaran, Natalie Asmus, Sahil Malhotra, Suraj Upadhya, Boris Kantor, Misha Angrist and Ornit Chiba-Falek

